#4 The Compound Eye
Pandemic response treaty, Mandatory vaccinations, Assisted Reproductive Technology (ART) Bill
Welcome old and new subscribers. This is the new version of the health and life-science policy newsletter started by Shambhavi Naik. We have new contributors on board. They are Priyal Lyncia D’Almeida, Harshit Kukreja, and Ruturaj Gowaikar. We have expanded our coverage to include Health systems and climate & environmental issues .
First step towards a global pandemic response treaty.
-Ruturaj Gowaikar @RuturajGowaikar
A special session of the World Health Assembly was held in November-December 2021.The agenda was to generate consensus towards a legally binding instrument amongst nations to ensure co-operation during global health emergencies. Dr Tedros Adhanom Ghebreyesus, WHO Director-General said that this was a once-in- a-generation opportunity to strengthen global health architecture. 25 heads of government and international agencies had given a joint call for this session.
The Need
The need for such a legally binding instrument arose from the reactive, knee-jerk measures that many countries implemented during the Sars-CoV-2 pandemic. Moreover, the WHO that had access to global data didn’t have the authority to declare a pandemic. The best it could do was declare a ‘public health emergency of international concern’ (PHEIC). A PHEIC doesn’t ensure global co-operation because the advisories issued under this framework are not legally binding. There was a communication gap between the WHO, head of states, the medical and research communities, and the public. Moreover, after vaccines were developed, countries resorted to ‘vaccine nationalism’ by hoarding vaccines in order to prioritise their own citizens. Countries were not forthcoming when it came to patent waivers to ramp-up vaccine production in other parts of the world either.
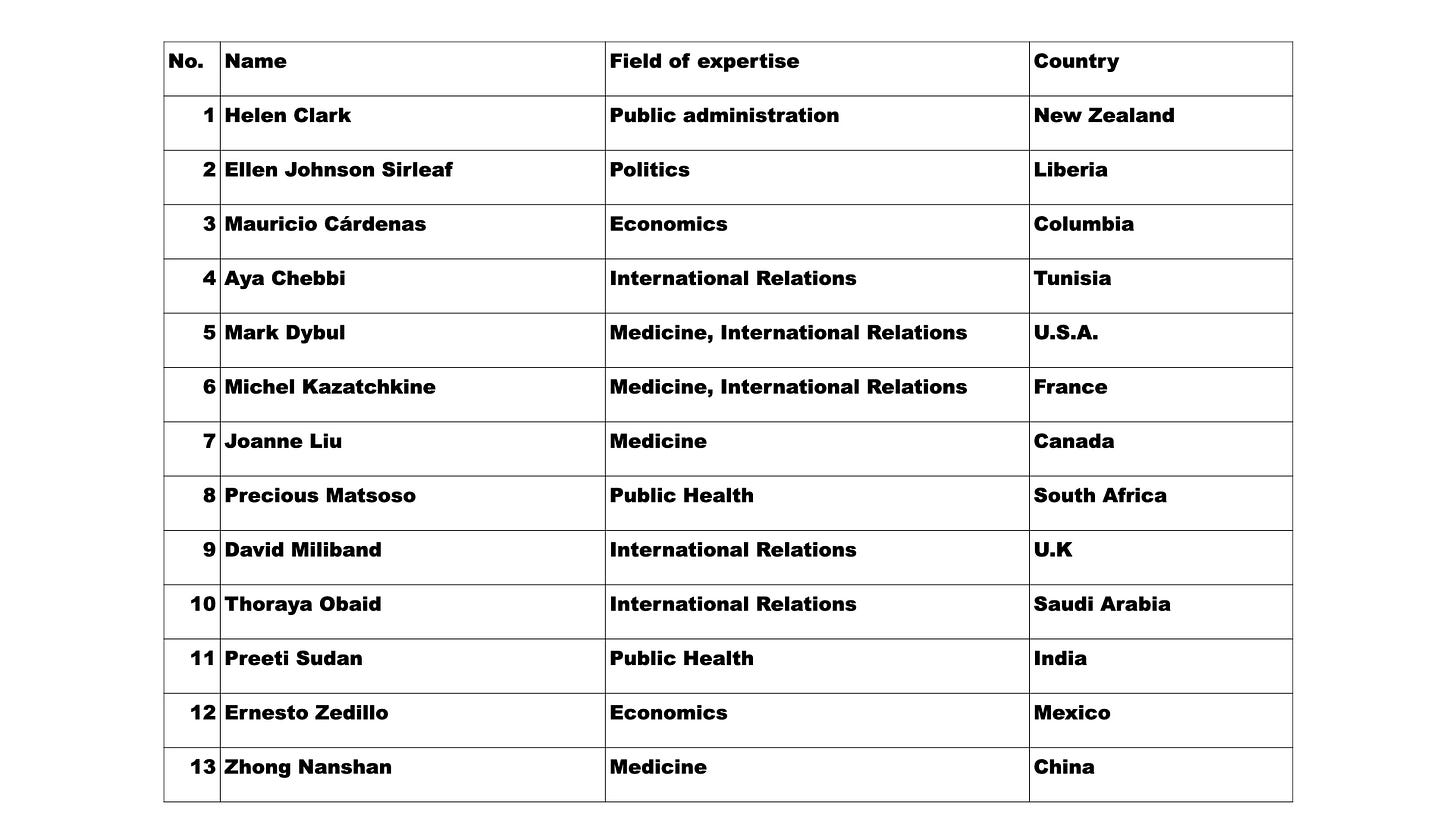
The WHO appointed a team of 13 independent experts to review the way Covid-19 pandemic unfolded and the global response to it. Their findings were presented as a report on 12th May 2021. Some of the recommendations are abstract in nature while there are some actionable suggestions to manage the current pandemic as well as future pandemics. One of the suggestions is to give more authority to the WHO via a legally binding instrument to achieve better coordination.
Following are the details of the experts.
Timelines
The special session in November-December 2021 was the 1st step towards building a consensus. This session saw countries committing to an idea of a draft of such an international legal framework. An intergovernmental negotiating body will now meet in March 2022 to start the process of writing a draft agreement and then in August 2022 to discuss the progress. The aim is to have a draft ready in time for the 76th World Assembly, and get countries to sign it by 2024.
Limited precedent
Such an international treaty is rare in the 74 years of existence of the WHO. The only time such an instrument was agreed upon was against an ‘epidemic’ of health issues related to tobacco products. It was called ‘WHO Framework Convention on Tobacco Control’ (WHO FCTC) and was adopted in 2013. This instrument was the first to address, demand reduction strategies as well as supply issues when it came to an addictive substance. The health warnings on tobacco products, ban on advertisements related to tobacco in any form, and age and geographical restrictions on sale of tobacco products are a result of this treaty.
Hits and Misses of the Special Health Assembly.
It was decided that the WHO needs to be given more authority in a pandemic like scenario and so modifications of International Health Regulations (IHRs) were suggested. IHRs is the international treaty that allows the WHO to be the leading agency for collecting global health data and issue advisories. It was also agreed upon that these new powers of the WHO would continue to adhere to article 19 of the constitution of the WHO. Article 19 is the article that bestows the World Health Assembly with the authority to adopt conventions or agreements on any matter within WHO’s competence.
The original call for this special session had ‘treaty’ in its declaration. But the U.S.A, Brazil, and India seemed unwilling to be subjected to this highest form of international legal instrument. The now agreed upon format is going to be an accord, a much less stringent instrument with a mechanism for nations to exit the instrument.
An intergovernmental negotiating body (INB) has been established that will work towards drafting this accord.What this accord will contain are robust mechanisms to share global health data and carrots and sticks for participating nations. These mechanisms can be similar to the Pandemic Influenza Preparedness (PIP) Framework which ensures vaccine access in lieu of data sharing.
What this accord might not contain are patent-waivers for vaccines or other medications. India and South Africa had brought up this issue before and during this special assembly as well but faced opposition from some EU countries. EU has some other mechanism to promote vaccine equity but not blanket patent-waivers.
Conclusion
The drafting of this accord and successful acceptance of it is a long process. Some of the clauses might seem utopian and too idealistic. But considering the severity of the Covid pandemic and success of the (WHO FCTC), this proposed new accord might be accepted and enable global cooperation for future pandemics.
Mandatory COVID Vaccinations: An Ethical Viewpoint
-Harshit Kukreja @harshitk43
With COVID 19 vaccinations being more readily available, we are finally inching towards a possible future where there are COVID 19 vaccinations for everybody. India has to start thinking about the people who are unwilling to get jabbed. This is called "Vaccine Hesitancy".
Is it ethical to limit an individual's liberty when their choice might harm others? This is the case with Mandatory COVID vaccines, where a person's choice of not getting a vaccine has negative repercussions for others.
Indian Situation
In India, as compared to the developed countries vaccine hesitancy is not very widely seen. Although, a majority are concerned about the adverse effects of the vaccine. The reasons vary from being unsure about the safety of the vaccines or a paucity of trust in modern medicine.
The COVID Vaccines currently in the market are quite safe. Around 90% of the vaccines given out in India are AstraZeneca's Covidshield. It has been deemed to be quite safe except for a few very rare adverse effects like a major blood clotting event affecting less than 1 per lakh. This risk can be deemed to be acceptable given the benefits.
Should the government force you to take this risk?
If we make COVID vaccinations compulsory for everybody or even for a subset of the population (for instance healthcare workers), the question arises that should the government decide for you whether the risks are acceptable or not.
In a Policy brief by WHO, WHO argues that deciding to make vaccinations mandatory or limiting individual choices when people refuse vaccinations is dependent on a few points. We look at them from an Indian context.
Necessity and proportionality: The Government should only issue a mandate if the population which is getting not getting vaccinated is being an impediment in achieving a public health goal (e.g., herd immunity, protecting the most vulnerable, protecting the capacity of the acute health care system). This should only be done after all other non-coercive measures have failed.
Sufficient evidence of vaccine safety: Indians have been getting vaccinated with mainly 2 vaccines: Covidshield and Covaxin. They have both been deemed to be quite safe with quite rare adverse effects. Even with these rare adverse effects, mandatory vaccines should be accompanied by schemes to compensate for any vaccine-related harm that might occur. This is cardinal and fair to compensate people for mandatory vaccine-related harm.
Sufficient evidence of vaccine efficacy and effectiveness- In India, with our huge population, large non- COVID 19 disease burden and our dismal healthcare system, currently available vaccines can be made mandatory for special populations. Although they are not 100% effective against preventing infections, they can significantly reduce severity and deaths.
Sufficient supply- "In order for a mandate to be considered, supply of the authorized vaccine should be sufficient and reliable, with reasonable, free access for those for whom it is to be made mandatory" Vaccines can be only mandated in our country after access is assured for the targeted population.
In Special Populations
Vaccines mandates are currently justified with their current efficacy, effectiveness and safety profile for populations such as the healthcare force. As they deal with the populations we want to protect( the immunocompromised, the elderly, the severely sick) and are quite vulnerable to infections. There is also added viewpoint of protecting the workforce that runs the healthcare system.
If measures like better communication and assurance of compensation in harm from vaccination fail, These mandates can be extended to populations that are essential and directly deal with the public such as teachers, policemen.
Finally when there is more data regarding vaccine safety, ways to mitigate vaccine harm and sufficient supply, we can think of a universal mandate for COVID-19 Vaccination.
Read More
https://www.healthaffairs.org/do/10.1377/forefront.20211029.682797/full/
https://www.who.int/publications/i/item/WHO-2019-nCoV-Policy-brief-Mandatory-vaccination-2021.1
https://www.nejm.org/doi/full/10.1056/NEJMp2115832
ART Bill: What is it? And what does it aim to do?
-Priyal Lyncia D’Almeida @LynciaPriyal
Parliament passed the Assisted Reproductive Technology (ART) Bill early last month during the winter session. This bill aims to regulate and supervise the assisted reproductive technology clinics by overseeing techniques such as the In-Vitro Fertilisation (IVF) and embryo transfer. Sperm banks are also added to the list of services that will be regulated by this bill, as this industry has seen a boom in recent years.
According to this bill,
The union government will appoint one or more appropriate assisted reproductive technology and surrogacy authorities. All clinics in India that offer ART services are to be registered under this authority. The bill also aims to establish a national Registry that will serve as a single database containing information on all the ART clinics and banks. To make the registration process easier, state governments will appoint registration authorities. These banks and clinics will only be granted the registration if they meet specific criteria such as trained staff, proper infrastructure and diagnostic facilities. This registration would be valid for five years, with an option to renew for another five years. If any clinic or bank violates any terms of this bill, their license/membership would be revoked or suspended. In the exercise of their powers and responsibilities. The National Registry, The National Board and The State Board holds the authority to visit any location relevant to ART and/or request any documents or papers.
Only a registered ART bank can screen gamete donors, collect and store sperm, and provide oocyte donors. Males between the ages of 21 and 55 can donate their sperm, while women between the ages of 23 and 55 can donate their oocytes. An oocyte donor should be a married lady with at least one child of three years or older. A female can contribute only one oocyte only once throughout her life and she can only have seven oocytes extracted from her. A single donor’s gamete cannot be sent to more than one commissioning couple by the bank.
ART services can only be performed when the person requesting ART services and the donor provide written consent. The party requiring ART treatments will be obliged to provide insurance to the oocyte donor incase of any loss, or damage or death of the donor. It is illegal for a any clinic to agree to give a kid’s predetermined sex. The bill also stipulates that before an embryo is implanted, it must be checked for genetic disorders.
The baby born via ART would be considered the biological child of the commissioning party. They will be entitled to all the rights and benefits that a natural child of the commissioning couple would have. The donor wouldn’t be able to exercise parental authority over the kid.
Multiple offenses have been included in this bill. Abandonment and exploitation of kids born through ART; Trading, importing, selling and purchasing of human embryos or gametes; trying to exploit the woman, gamete donor or the commissioning couple in any form; and transferring the human embryo into a male or animals are all considered criminal offences. For the first violation, a fine of five to ten lakhs would be charged. For repeated violations, a fine of anywhere between ten to fifteen lakhs would be charged while also being punished by imprisonment for eight to twelve years. Any clinic or bank that advertises or offers gender-selective ART would face a sentence for five to ten years in prison or a penalty of rupees ten to twenty five lakhs, or both.
Despite the bill being a great step ahead in regulating the ART clinics and facilities in India, we need to address the fact that this bill discriminates against the LGBTQIA+ people, couples in live-in relationships, and single male parents. This was also pointed out during the Lok Sabha discussion. At its heart, the statute contradicts the supreme court’s ruling by perpetuating discrimination against same sex couples in their ability to raise a family. In this respect, it appears to be that the ART bill was influenced by the heteronormative beliefs. This gets reiterated as the ART bill defines the commissioning couple as an “infertile married couple”.
While the bill definitely aims to address and reduce a huge lacuna in the nation’s reproductive technology laws, the government must uphold its constitutional commitment to treat all people equally and fix sections that ultimately set up classifications that have no rational nexus with the goal.